Upload time:2020-12-30| Author:admin
Lithium metal batteries generally use manganese dioxide as cathode material, lithium metal or its alloy metal as anode material, and non-aqueous electrolyte solution.
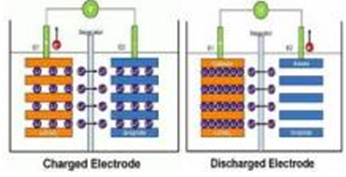
Basic principle of lithium battery
Discharge reaction: Li + MnO2 = LiMnO2
Lithium ion battery:
Lithium ion batteries generally use lithium alloy metal oxide as cathode material, graphite as anode material and non-aqueous electrolyte.
The reaction on the positive electrode of charging is
LiCoO2 = = Li (1-x) coo2 + XLI + + XE - (electron)
The reaction on the negative electrode is
6C+XLi++Xe- = LixC6
Total reaction of rechargeable battery: LiCoO2 + 6C = Li (1-x) coo2 + lixc6
Positive electrode
Cathode material: there are many optional cathode materials, and lithium iron phosphate is widely used in mainstream products. Comparison of different cathode materials:
|
LiCoO2 |
3.7 V |
140 mAh/g |
|
Li2Mn2O4 |
4.0 V |
100 mAh/g |
|
LiFePO4 |
3.3 V |
100 mAh/g |
|
Li2FePO4F |
3.6 V |
115 mAh/g |
Positive reaction: lithium ion is embedded during discharge, and lithium ion is de embedded during charging. Charging: LiFePO4 → li1-xfepo4 + XLI + + XE - discharging: li1-xfepo4 + XLI + + XE - → LiFePO4.
Negative pole
Anode material: graphite. New research has found that titanate may be a better material.
Negative electrode reaction: Li ion is de intercalated during discharge and Li ion is intercalated during charging.
Charging: XLI + + XE - + 6C → lixc6
During discharge: lixc6 → XLI + + XE - + 6C
18038100011
Phone:13530889719
Tel:0755-33564871
Fax:0755-27850441
E-mail:sales@cxytech.com
Address: No. 501, 5th Floor, Block A, Huafeng Internet Creative Park, No. 107, Gonghe Industrial Road, Xixiang Street, Baoan District, Shenzhen
Dongguan Factory Address: 3rd Floor, Building B, Huazhao Technology Park, No. 8 Xinhui Road, Xin'an Community, Chang'an Town, Dongguan City

Mobile QR code